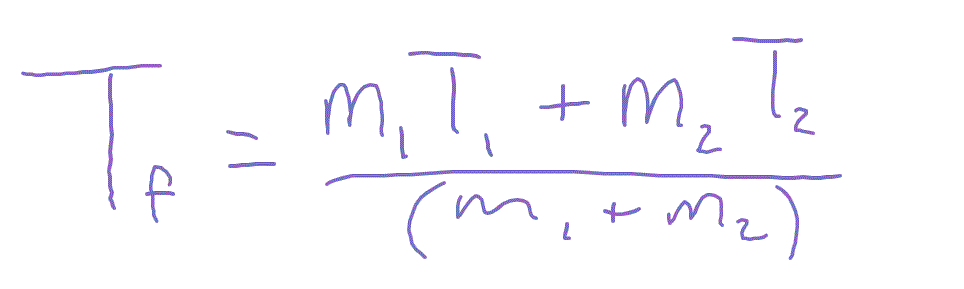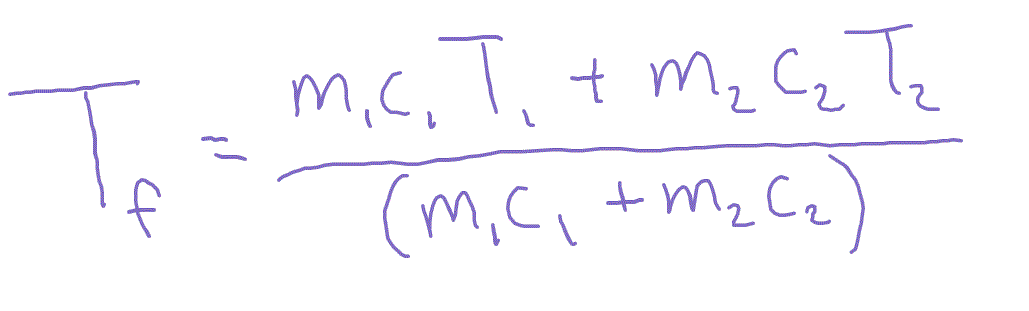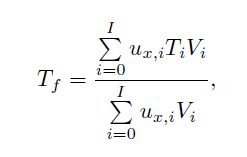50.0mL of 0.10 M HCl is mixed with 50.0mL of 0.10 M NaOH .The solution temperature rises by 3.0^(@)CCalculate

SOLVED:0 23 27 based on the information blow temperature ot 25'€ The treated wastcwater biG35 wastcwater treatment plant at 20,000 m"/d is treated ateleased In 3 river having the flow Tate o/
2 kg of water at 0° c is mixed with the same amount of water at 60° c. What is the change in entropy? - Quora

coupled tank system mixed water system for wall mounted furnace mixed water temperature calculation SS304 100w/165w|Floor Heating Systems & Parts| - AliExpress
Bounding Estimate for the 'Hot' Channel Temperature and Preliminary Calculation of Mixing in the Lower Plenum for the NGNP Point Design Using CFD - UNT Digital Library

A 18.7-g sample of ice at -13.1°C is mixed with 118.5 g of water at 80.0°C. Calculate the final temperature of the mixtu... - HomeworkLib