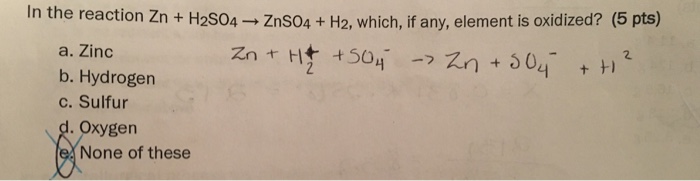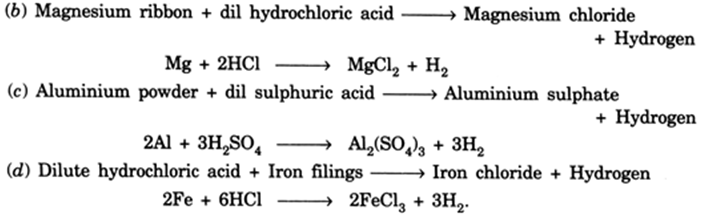
Write word equations and then balanced equations for the reaction taking place when:(a) dilute sulphuric acid reacts with zinc granules.(b) dilute hydrochloric acid reacts with magnesium ribbon.(c) dilute sulphuric acid reacts with

Q 3 The reaction is given by Zn+H2SO4→ZnSO4+H2 (i) White the ionic equation for the reaction (ii) - Science - Chemical Reactions and Equations - 12384659 | Meritnation.com
Balance the following equation by oxidation number method : (i) K2Cr2O7 + KCl + H2SO4 → KHSO4 + CrO2Cl2 + H2O - Sarthaks eConnect | Largest Online Education Community

OneClass: An impure sample of zinc (Zn) is treated with an excess of sulfuric acid (H2SO4) to form zi...
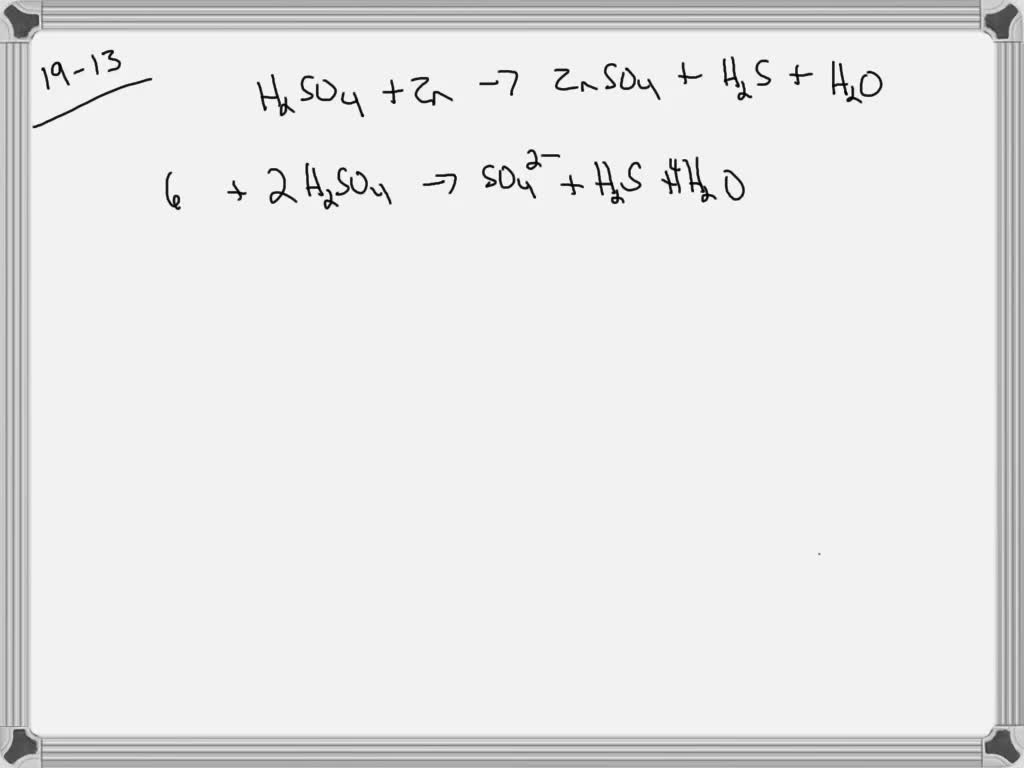
SOLVED:Balance the equation for the reaction in which hot, concentrated sulfuric acid reacts with zinc to form zinc sulfate, hydrogen sulfide, and water.

The reaction is given by Zn+h2so4 gives znso4+h2 The ionic equations can be represented by two half - Brainly.in

Which of the following is the correct cell representation for the given cell reaction? Zn + H2SO4→ ZnSO4 + H2
Consider the following reactions (unbalanced) Zn + hot conc. H2SO4 →G + R + X - Sarthaks eConnect | Largest Online Education Community